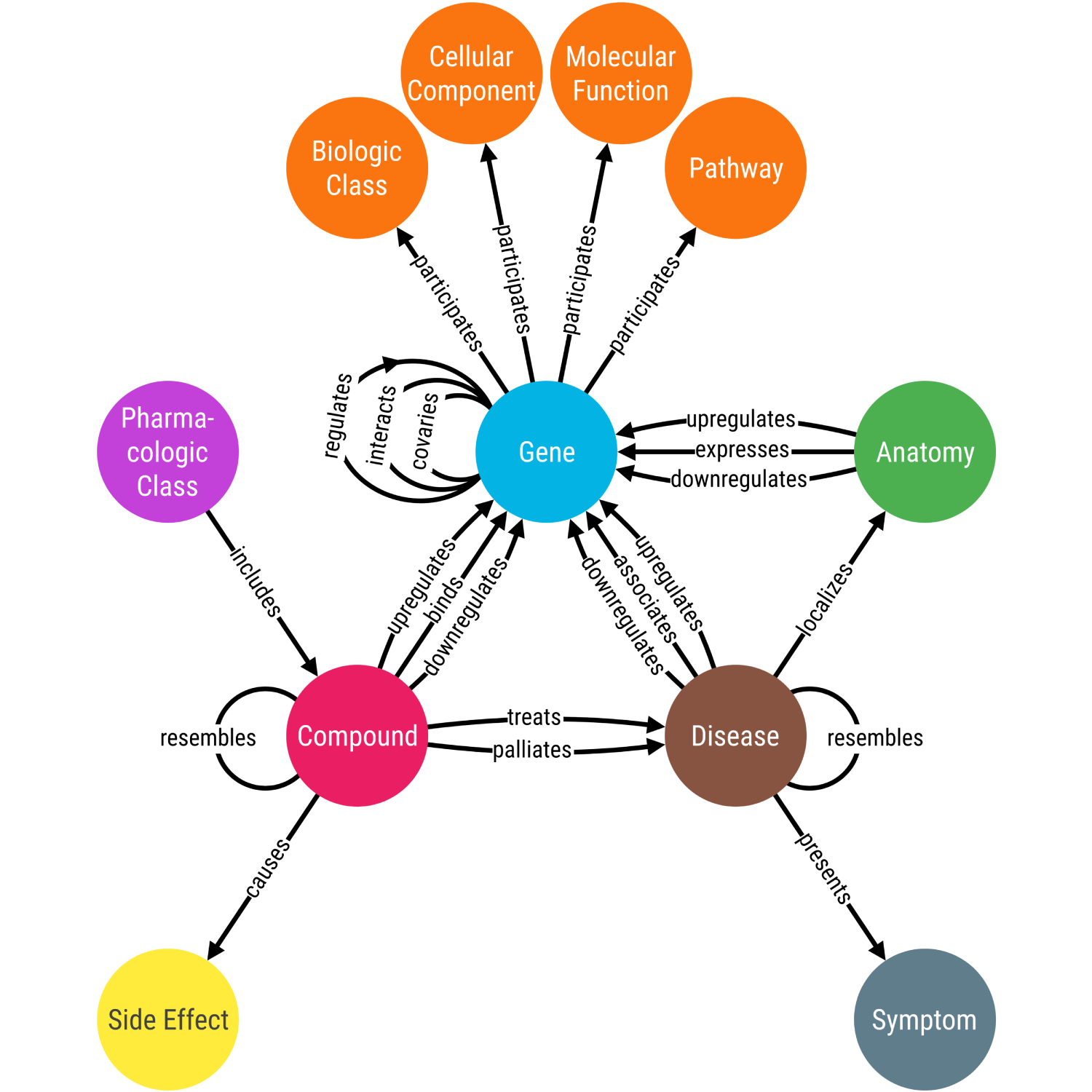
Knowledge graphs can support many biomedical applications.
These graphs represent biomedical concepts and relationships in the form of nodes and edges.
In this review, we discuss how these graphs are constructed and applied with a particular focus on how machine learning approaches are changing these processes.
Biomedical knowledge graphs have often been constructed by integrating databases that were populated by experts via manual curation, but we are now seeing a more robust use of automated systems.
A number of techniques are used to represent knowledge graphs, but often machine learning methods are used to construct a low-dimensional representation that can support many different applications.
This representation is designed to preserve a knowledge graph’s local and/or global structure.
Additional machine learning methods can be applied to this representation to make predictions within genomic, pharmaceutical, and clinical domains.
We frame our discussion first around knowledge graph construction and then around unifying representational learning techniques and unifying applications.
Advances in machine learning for biomedicine are creating new opportunities across many domains, and we note potential avenues for future work with knowledge graphs that appear particularly promising.
Graphs are practical resources for many real-world applications.
They have been used in social network mining to classify nodes [1] and create recommendation systems [2].
They have also been used in natural language processing to interpret simple questions and use relational information to provide answers [3,4].
In a biomedical setting, graphs have been used to prioritize genes relevant to disease [5,6,7,8], perform drug repurposing [9] and identify drug-target interactions [10].
Within a biomedical setting, some graphs can be considered knowledge graphs; although, precisely defining a knowledge graph is difficult because there are multiple conflicting definitions [11].
For this review, we define a biomedical knowledge graph as the following: a resource that integrates one or more expert-derived sources of information into a graph where nodes represent biomedical entities and edges represent relationships between two entities.
This definition is consistent with other definitions found in the literature [12,13,14,15,16,17,18].
Often relationships are considered unidirectional (e.g., a compound treats a disease, but a disease cannot treat a compound); however, there are cases where relationships can be considered bidirectional (e.g., a compound resembles another compound, or a gene interacts with another gene).
A subset of graphs that meet our definition of a knowledge graph would be unsuitable for applications such as symbolic reasoning [19]; however, we chose a more liberal definition because it has been demonstrated that these broadly defined graphs have numerous uses throughout the literature.
For example, Hetionet (Figure 1) [9] would be considered a biomedical knowledge graph by this definition, and it has been used to identify drug repurposing opportunities [9].
We do not consider databases like DISEASES [20] and DrugBank [21] to be knowledge graphs.
Although these resources contain essential information, they do not represent their data in the form of a graph.
Biomedical knowledge graphs are often constructed from manually curated databases [9,10,22,23,24].
These databases provide previously established information that can be incorporated into a graph.
For example, a graph using DISEASES [20] as a resource would have genes and diseases as nodes, while edges added between nodes would represent an association between a gene and a disease.
This example shows a single type of relationship; however, there are graphs that use databases with multiple relationships [9,25].
In addition to manual curation, other approaches have used natural language processing techniques to construct knowledge graphs [26,27].
One example used a text mining system to extract sentences that illustrate a protein’s interaction with another protein [28].
Once identified, these sentences can be incorporated as evidence to establish an edge in a knowledge graph.
In this review we describe various approaches for constructing and applying knowledge graphs in a biomedical setting.
We discuss the pros and cons of constructing a knowledge graph via manually curated databases and via text mining systems.
We also compare assorted approaches for applying knowledge graphs to solve biomedical problems.
Lastly, we conclude on the practicality of knowledge graphs and point out future applications that have yet to be explored.
Knowledge graphs can be constructed in many ways using resources such as pre-existing databases or text.
Usually, knowledge graphs are constructed using pre-existing databases.
These databases are constructed by domain experts using approaches ranging from manual curation to automated techniques, such as text mining.
Manual curation is a time-consuming process that requires domain experts to read papers and annotate sentences that assert a relationship.
Automated approaches rely on machine learning or natural language processing techniques to rapidly detect sentences of interest.
We categorize these automated approaches into the following groups: rule-based extraction, unsupervised machine learning, and supervised machine learning and discuss examples of each type of approach while synthesizing their strengths and weaknesses.
Database construction dates back all the way to 1956 when the first database contained a protein sequence of the insulin molecule [29].
The process of database construction involves gathering relevant text such as journal articles, abstracts, or web-based text and having curators read the gathered text to detect sentences that implicate a relationship (i.e., relationship extraction).
Notable databases constructed by this process can be in found in Table 1.
An example database, COSMIC [30] was constructed by a group of domain experts scanning the literature for key cancer related genes.
This database contained approximately 35M entries in 2016 [30] and by 2018 had grown to 45M entries [31].
Studies have shown that databases constructed in this fashion contain relatively precise data but the recall is low [32,33,34,35,36,37,38].
Low recall happens because the publication rate is too high for curators to keep up [39].
This bottleneck highlights a critical need for future approaches to scale fast enough to compete with the increasing publication rate.
Despite the negatives of manual curation, it is still an essential process for extracting relationships from text.
This process can be used to generate gold standard datasets that automated systems use for validation [49,50] and can be used during the training process of these systems (i.e., active learning) [51].
It is important to remember that manual curation alone is precise but results in low recall rates [38].
Future databases should consider initially relying on automated methods to obtain sentences at an acceptable recall level, then incorporate manual curation as a way to fix or remove irrelevant results.
Rule-based extraction consists of identifying essential keywords and grammatical patterns to detect relationships of interest.
Keywords are established via expert knowledge or through the use of pre-existing ontologies, while grammatical patterns are constructed via experts curating parse trees.
Parse trees are tree data structures that depict a sentence’s grammatical structure and come in two forms: a constituency parse tree (Figure 2) and a dependency parse tree (Figure 3).
Both trees use part of speech tags, labels that dictate the grammatical role of a word such as noun, verb, adjective, etc., for construction, but represent the information in two different forms.
Constituency parse trees break a sentence into subphrases (Figure 2) while dependency path trees analyze the grammatical structure of a sentence (Figure 3).
Many text mining approaches [59,60,61] use such trees to generate features for machine learning algorithms and these approaches are discussed in later sections.
In this section we focus on approaches that use rule-based extraction as a primary strategy to detect sentences that allude to a relationship.
Pattern matching is a fundamental approach used to detect relationship asserting sentences.
These patterns can consist of phrases from constituency trees, a set of keywords or some combination of both [36,67,68,69,70,71].
Xu et al. designed a pattern matcher system to detect sentences in PubMed abstracts that indicate drug-disease treatments [70].
This system matched drug-disease pairs from ClinicalTrials.gov to drug-disease pairs mentioned in abstracts.
This matching process aided the authors in identifying sentences that can be used to create simple patterns, such as “Drug in the treatment of Disease” [70], to match other sentences in a wide variety of abstracts.
The authors hand curated two datasets for evaluation and achieved a high precision score of 0.904 and a low recall score of 0.131 [70].
This low recall score was based on constructed patterns being too specific to detect infrequent drug pairs.
Besides constituency trees, some approaches used dependency trees to construct patterns [59,72].
Depending upon the nature of the algorithm and text, dependency trees could be more appropriate than constituency trees and vice versa.
The performance difference between the two trees remains as an open question for future exploration.
Rule-based methods provide a basis for many relationship extraction systems.
Approaches in this category range from simplifying sentences for easy extraction to identifying sentences based on matched key phrases or grammatical patterns.
Both require a significant amount of manual effort and expert knowledge to perform well.
A future direction is to develop ways to automate the construction of these hand-crafted patterns, which would accelerate the process of creating these rule-based systems.
Unsupervised extractors draw inferences from textual data without the use of annotated labels.
These methods involve some form of clustering or statistical calculations.
In this section we focus on methods that use unsupervised learning to extract relationships from text.
An unsupervised extractor can exploit the fact that two entities may appear together in text.
This event is referred to as co-occurrence and studies that use this phenomenon can be found in Table 2.
Two databases DISEASES [20] and STRING [75] were populated using a co-occurrence scoring method on PubMed abstracts, which measured the frequency of co-mention pairs within individual sentences as well as the abstracts themselves.
This technique assumes that each individual co-occurring pair is independent from one another.
Under this assumption mention pairs that occur more than expected were presumed to implicate the presence of an association or interaction.
This approach identified 543,405 disease gene associations [20] and 792,730 high confidence protein-protein interactions [75] but is limited to only PubMed abstracts.
Unsupervised extractors often treat different biomedical relationships as multiple isolated problems.
An alternative to this perspective is to capture all different types at once.
Clustering is an approach that performs simultaneous extraction.
Percha et al. used a biclustering algorithm on generated dependency parse trees to group sentences within PubMed abstracts [78].
Each cluster was manually curated to determine which relationship each group represented.
This approach captured 4,451,661 dependency paths for 36 different groups [78].
Despite the success, this approach suffered from technical issues such as dependency tree parsing errors.
These errors resulted in some sentences not being captured by the clustering algorithm [78].
Future clustering approaches should consider simplifying sentences to prevent this type of issue.
Overall unsupervised methods provide a means to rapidly extract relationship asserting sentences without the need of annotated text.
Approaches in this category range from calculating co-occurrence scores to clustering sentences and provide a generalizable framework that can be used on large repositories of text.
Full text has already been shown to meaningfully improve the performance of methods that aim to infer relationships using cooccurrences [76], and we should expect similar benefits for machine learning approaches.
Furthermore, we expect that simplifying sentences would improve unsupervised methods and should be considered as an initial preprocessing step.
Supervised extractors use labeled sentences to construct generalized patterns that bisect positive examples (sentences that allude to a relationship) from negative ones (sentences that do not allude to a relationship).
Most of these approaches have flourished due to pre-labelled publicly available datasets (Table 3).
These datasets were constructed by curators for shared open tasks [84,85] or as a means to provide the scientific community with a gold standard [85,86,87].
Approaches that use these available datasets range from using linear classifiers such as support vector machines (SVMs) to non-linear classifiers such as deep learning techniques.
The rest of this section discusses approaches that use supervised extractors to detect relationship asserting sentences.
Some supervised extractors involve the mapping of textual input into a high dimensional space.
SVMs are a type of classifier that can accomplish this task with a mapping function called a kernel [61,88].
These kernels take information such as a sentence’s dependency tree [59,60], part of speech tags [61] or even word counts [88] and map them onto a dense feature space.
Within this space, these methods construct a hyperplane that separates sentences in the positive class (illustrates a relationship) from the negative class (does not illustrate a relationship).
Kernels can be manually constructed or selected to cater to the relationship of interest [60,61,88,88].
Determining the correct kernel is a nontrivial task that requires expert knowledge to be successful.
In addition to single kernel methods, a recent study used an ensemble of SVMs to extract disease-gene associations [89].
This ensemble outperformed notable disease-gene association extractors [72,90] in terms of precision, recall and F1 score.
Overall, SVMs have been shown to be beneficial in terms of relationship mining; however, major focus has shifted to utilizing deep learning techniques which can perform non-linear mappings of high dimensional data.
Deep learning is an increasingly popular class of techniques that can construct their own features within a high dimensional space [91,92].
These methods use different forms of neural networks, such as recurrent or convolutional neural networks, to perform classification.
Recurrent neural networks (RNN) are designed for sequential analysis and use a repeatedly updating hidden state to make predictions.
An example of a recurrent neural network is a long short-term memory (LSTM) network [93].
Cocos et al. [94] used a LSTM to extract drug side effects from de-identified twitter posts, while Yadav et al. [???] used an LSTM to extract protein-protein interactions.
Others have also embraced LSTMs to perform relationship extraction [94,95,96,97,98].
Despite the success of these networks, training can be difficult as these networks are highly susceptible to vanishing and exploding gradients [99,100].
One proposed solution to this problem is to clip the gradients while the neural network trains [101].
Besides the gradient problem, these approaches only peak in performance when the datasets reach at least tens of thousands of data points [102].
Convolutional neural networks (CNNs), which are widely applied for image analysis, use multiple kernel filters to capture small subsets of an overall image [92].
In the context of text mining an image is replaced with words within a sentence mapped to dense vectors (i.e., word embeddings) [103,104].
Peng et al. used a CNN to extract sentences that mentioned protein-protein interactions [105] and Zhou et al. used a CNN to extract chemical-disease relations [106].
Others have used CNNs and variants of CNNs to extract relationships from text [107,108,109].
Just like RNNs, these networks perform well when millions of labeled examples are present [102]; however, obtaining these large datasets is a non-trivial task.
Future approaches that use CNNs or RNNs should consider solutions to obtaining these large quantities of data through means such as weak supervision [110], semi-supervised learning [111] or using pre-trained networks via transfer learning [112,113].
Weak or distant supervision takes a different approach by using noisy or even erroneous labels to train classifiers [110,115,116,117].
Under this paradigm, sentences are labeled based on their mention pair being present (positive) or absent (negative) in a database and, once labeled, a machine learning classifier can be trained to extract relationships from text [110].
For example, Thomas et al. [118] used distant supervision to train a SVM to extract sentences mentioning protein-protein interactions (PPI).
Their SVM model achieved comparable performance against a baseline model; however, the noise generated via distant supervision was difficult to eradicate [118].
A number of efforts have focused on combining distant supervision with other types of labeling strategies to mitigate the negative impacts of noisy knowledge bases [119,120,121].
Nicholson et al. [109] found that, in some circumstances, these strategies can be reused across different types of biomedical relationships to learn a heterogeneous knowledge graph in cases where those relationships describe similar physical concepts.
Combining distant supervision with other types of labeling strategies remains an active area of investigation with numerous associated challenges and opportunities.
Overall, semi-supervised learning and weak supervision provide promising results in terms of relationship extraction and future approaches should consider using these paradigms to train machine learning classifiers.
Knowledge graphs can help researchers tackle many biomedical problems such as finding new treatments for existing drugs [9], aiding efforts to diagnose patients [126] and identifying associations between diseases and biomolecules [127].
In many cases, solutions rely on representing knowledge graphs in a low dimensional space, which is a process called representational learning.
The goal of this process is to retain and encode the local and/or global structure of a knowledge graph that is relevant to the problem while transforming the graph into a representation that can be readily used with machine learning methods to build predictors.
In the following sections we review methods that construct a low dimensional space (Unifying Representational Learning Techniques) and discuss applications that use this space to solve biomedical problems (Unifying Applications).
Mapping high dimensional data into a low dimensional space greatly improves modeling performance in fields such as natural language processing [103,104] and image analysis [128].
The success of these approaches served as rationale for a sharper focus on representing knowledge graphs in a low dimensional space [129].
Methods of this class are designed to capture the essence of a knowledge graph in the form of dense vectors [130,131].
These vectors are often assigned to nodes in a graph [132], but edges can be assigned as well [133].
Techniques that construct a low dimensional space often require information on how nodes are connected with one another [134,135,136,137], while other approaches can work directly with the edges themselves [138].
Once this space has been constructed, machine learning techniques can utilize the space for downstream analyses such as classification or clustering.
We group techniques that construct this space into the following three categories: matrix factorization, translational distance models, and neural network models (Figure 4).
Matrix factorization is a class of techniques that use linear algebra to map high dimensional data into a low dimensional space.
This projection is accomplished by decomposing a matrix into a set of small rectangular matrices (Figure 4 (a)).
Notable methods for matrix decomposition include Isomap [139], Laplacian eigenmaps [131] and Principal Component Analysis (PCA) [140]/Singular Vector Decomposition (SVD) [130].
These methods were designed to be used on many different types of data; however, we discuss their use in the context of representing knowledge graphs in a low dimensional space and focus particularly on SVD and laplacian eigenmaps.
Laplacian eigenmaps assume there is low dimensional structure in a high dimensional space and preserves this structure when projecting data into a low dimensional space [131].
The first step of this technique is to preserve the low dimensional structure by representing data in the form of a graph where nodes are datapoints and edges are the distance between two points.
Knowledge graphs already provide this representation, so no additional processing is necessary at this stage.
The second step of this technique is to obtain both an adjacency matrix (\(A\)) and a degree matrix (\(D\)) from the graph representation.
A degree matrix is a diagonal matrix where each entry represents the number of edges connected to a node.
The adjacency and degree matrices are converted into a laplacian matrix (\(L\)), which is a matrix that shares the same properties as the adjacency matrix.
The laplacian matrix is generated by subtracting the adjacency matrix from the degree matrix (\(L=D-A\)) and, once constructed, the algorithm uses linear algebra to calculate the laplacian’s eigenvalues and eigenvectors (\(Lx = \lambda Dx\)).
The generated eigenvectors represent the knowledge graph’s nodes represented in a low dimensional space [131].
Other efforts have used variants of this algorithm to construct low dimensional representations of knowledge graphs [134,135,144].
Typically, eigenmaps work well when knowledge graphs have a sparse number of edges between nodes but struggle when presented with denser networks [141,144,145].
An open area of exploration is to adapt these methods to accommodate knowledge graphs that have a large number of edges.
Matrix factorization is a powerful technique that represents high dimensional data in a low dimensional space.
The representation of a knowledge graph in this reduced space does not meet our definition of a knowledge graph; however, this representation supports many use cases including similarity-based (e.g., cosine similarity [146]) and machine learning applications.
Common matrix factorization approaches involve using SVD, Laplacian eigenmaps or variants of the two to decompose matrices into smaller rectangular forms.
Regarding knowledge graphs, the adjacency matrix (\(A\)) is the typical matrix that gets decomposed, but the laplacian matrix (\(L=D-A\)) can be used as well.
Despite reported success, the dependence on matrices creates an issue of scalability as matrices of large networks may reach memory limitations.
Furthermore, the approaches we discussed consider all edge types as equivalent.
These limitations could be mitigated by new approaches designed to accommodate multiple node and edge types separately.
Translational distance models treat edges in a knowledge graph as linear transformations.
For example, one such algorithm, TransE [133], treats every node-edge pair as a triplet with head nodes represented as \(\textbf{h}\), edges represented as \(\textbf{r}\), and tail nodes represented as \(\textbf{t}\).
These representations are combined into an equation that mimics the iconic word vectors translations (\(\textbf{king} - \textbf{man} + \textbf{woman} \approx \textbf{queen}\)) from the word2vec model [104].
The described equation is shown as follows: \(\textbf{h} + \textbf{r} \approx \textbf{t}\).
Starting at the head node (\(\textbf{h}\)), one adds the edge vector (\(\textbf{r}\)) and the result should be the tail node (\(\textbf{t}\)).
TransE optimizes vectors for \(\textbf{h}\), \(\textbf{r}\), \(\textbf{t}\), while guaranteeing the global equation (\(\textbf{h} + \textbf{r} \approx \textbf{t}\)) is satisfied [133].
A caveat to the TransE approach is that it forces relationships to have a one to one mapping, which may not be appropriate for all relationship types.
Wang et al. attempted to resolve the one to one mapping issue by developing the TransH model [147].
TransH treats relations as hyperplanes rather than a regular vector and projects the head (\(\textbf{h}\)) and tail (\(\textbf{t}\)) nodes onto a hyperplane.
Following this projection, a distance vector (\(\textbf{d}_{r}\)) is calculated between the projected head and tail nodes.
Finally, each vector is optimized while preserving the global equation: \(\textbf{h} + \textbf{d}_{r} \approx \textbf{t}\) [147].
Other efforts have built off of the TransE and TransH models [148,149].
In the future, it may be beneficial for these models to incorporate other types of information such as edge confidence scores, textual information, or edge type information when optimizing these distance models.
Neural networks are a class of machine learning models inspired by the concept of biological neural networks [150].
These networks are reputable for making non-linear transformations of high dimensional data to solve classification and regression problems [150].
In the context of knowledge graphs, the most commonly used structures are based on word2vec [103,104].
The word2vec term applies to a set of conceptually related approaches that are widely used in the natural language processing field.
The goal of word2vec is to project words onto a low dimensional space that preserves their semantic meaning.
Strategies for training word2vec models use one of two neural network architectures: skip-gram and continuous bag of words (CBOW).
Both models are feed-forward neural networks, but CBOW models are trained to predict a word given its context while skip-gram models are trained to predict the context given a word [103,104].
Once training is completed, words will be associated with dense vectors that downstream models, such as feed forward networks or recurrent networks, can use for input.
Deepwalk is an early method that represents knowledge graphs in a low dimensional space [151].
The first step of this method is to perform a random walk along a knowledge graph.
During the random walk, every generated sequence of nodes is recorded and treated as a sentence in word2vec [103,104].
After every node has been processed, a skip-gram model is trained to predict the context of each node thereby constructing a low dimensional representation of a knowledge graph [151].
A limitation for deepwalk is that the random walk cannot be controlled, so every node has an equal chance to be reached.
Grover and Leskovec demonstrated that this limitation can hurt performance when classifying edges between nodes and developed node2vec as a result [132].
Node2vec operates in the same fashion as deepwalk; however, this algorithm specifies a parameter that lets the random walk be biased when traversing nodes [132].
A caveat to both deepwalk and node2vec is that they ignore information such as edge type and node type.
Various approaches have evolved to fix this limitation by incorporating node, edge and even path types when representing knowledge graphs in a low dimensional space [152,153,154,155].
An emerging area of work is to develop approaches that capture both the local and global structure of a graph when constructing this low dimensional space.
Though word2vec is the most common framework used to represent graphs, neural networks are sometimes designed to use the adjacency matrix as input [103,104].
These approaches use models called autoencoders [156,157,158].
Autoencoders are designed to map input into a low dimensional space and then back to a reconstruction of the same input [159,160].
It is possible to layer on additional objectives by modifying the loss function to take into account criteria above and beyond reconstruction loss [161,162].
In the context of knowledge graphs, the generated space correlates nodes with dense vectors that capture a graph’s connectivity structure [156,157,158].
Despite the high potential of autoencoders, this method relies on an adjacency matrix for input which can run into scalability issues as a knowledge graph asymptotically increases in size [163].
Plus, Khosla et al. discovered that approaches akin to node2vec outperformed algorithms using autoencoders when undergoing link prediction and node classification [163].
Overall, the performance of neural network models largely depends upon the structure of nodes and edges within a knowledge graph [163].
Furthermore, when these approaches are used only nodes are explicitly represented by these vectors.
This means a represented knowledge graph no longer meets our definition of a knowledge graph; however, this representation can make it more suitable for many biomedical applications.
Future areas of exploration should include hybrid models that use both node2vec and autoencoders to construct complementary low dimensional representations of knowledge graphs.
Knowledge graphs have been applied to many biomedical challenges ranging from identifying proteins’ functions [164] to prioritizing cancer genes [165] to recommending safer drugs for patients [166,167] (Figure 5).
In this section we review how knowledge graphs are applied in biomedical settings and put particular emphasis on an emerging set of techniques that represent knowledge graphs in a low dimensional space.
Multi-omic applications employ knowledge graphs to study the genome, how genes are expressed in the transcriptome, and how the products of those transcripts interact in the proteome.
These graphs are used to establish connections between -omic entities as well as diseases.
Tasks in this context include gene-symptom prioritization [168], protein-protein interaction prediction [169,170] and detecting miRNA-disease associations [127].
We focus specifically on multi-omic applications that represent knowledge graphs in a low dimensional space to make connections.
Recommendation systems make use of knowledge graphs to establish links between RNA with disease and proteins with other proteins.
Shen et al. used an algorithm called collaborative filtering to establish an association between miRNA and diseases [127].
The authors constructed a miRNA-Disease network using the Human MicroRNA Disease database (HMDD) [171] and generated an adjacency matrix with the rows representing miRNA and the columns representing diseases.
This matrix was decomposed into small rectangular matrices using SVD, then these small matrices were used to calculate similarity scores between miRNAs and diseases.
High scores implied a high likelihood that a given miRNA had an association with a given disease [127].
Other approaches built off of Shen et al.’s work by incorporating novel ways to perform matrix factorization [172,173,174] or by integrating machine learning models in conjunction with matrix factorization [175].
These approaches achieved high area under the receiver operating curve (AUROC), but new discoveries have been hard to validate as experiments in this space are costly and time consuming at best [127].
Apart from miRNA, collaborative filtering has been used to predict protein-protein interactions [169,170,176].
Although extensive validation of newly generated candidates may be impractical, it would be helpful to see future efforts in this space include a blinded literature search for prioritized and randomly selected candidates as part of the standard evaluation pipeline.
Knowledge graphs benefited the multi-omics field as a resource for generating novel discoveries.
Most approaches to date use matrix factorization and node2vec to project knowledge graph into a low dimensional space, while translational models (Figure 4 (b)) may be an untapped resource that could aid future efforts.
Another area of exploration could be incorporating multiple sources of information such as compounds, anatomic locations or genetic pathways to improve the specificity of findings (i.e., to predict that a protein-protein interaction happens in a specific cell type or tissue).
There are a multitude of examples where knowledge graphs have been applied to identify new properties of drugs.
Tasks in this field involve predicting drugs interacting with other drugs [183], identifying molecular targets a drug might interact with [184] and identifying new disease treatments for previously established drugs [185].
In this section we concentrate on applications that apply these graphs to discover new properties of drugs and focus on approaches that use these graphs in a low-dimensional space.
Similar to multi-omic applications, recommendation systems have utilized knowledge graphs to infer novel links between drugs and diseases.
Dai et al. used collaborative filtering to infer drug-disease associations [184].
The authors constructed a drug-disease network by integrating two bipartite networks: a drug-gene interaction network and a disease-gene interaction network.
They integrated both networks under the assumption that drugs associated with a disease interact with the same gene of interest.
Following construction, the authors generated an adjacency matrix where rows represent drugs and columns represent diseases.
This matrix was decomposed into two small rectangular matrices and these matrices were used to calculate similarity scores between all drugs and all diseases.
High values implied a high chance of an association [184].
Related approaches used this technique to infer drug-target interactions [186,187,188] and drug-disease treatments [189,190,191,192,193].
In spite of reported success, these approaches are limited to the drugs and diseases contained in the graph.
Combining these approaches with representations of chemical structures might make it possible to one day make predictions about novel compounds.
Applications that discover new properties of drugs have benefited from using knowledge graphs as a resource.
Most methods to date use matrix factorization and neural network models to produce a low-dimensional representation.
Due to the success of neural networks [200,201] much of the field’s focus has shifted to these techniques; however, a possible improvement is to use an ensemble of neural network models and linear methods to improve performance.
Another potential avenue for future work would be to incorporate entity-specific hierarchical information or similarity information to improve detection power.
For drugs, this could include pharmaceutical classes or chemical structure similarities.
Clinical applications that use knowledge graphs are in early stages of development, but the long-term goal is to use analyses of these graphs to aid patient care.
Typically, graphs for these applications are constructed from electronic health records (EHR): nodes represent patients, drugs and diseases while edges represent relationships such as a patient being prescribed a treatment or a patient being diagnosed with a disease [26,202,203,204].
Tasks within this field range from improving patient diagnoses [205,206] to recommending safer drugs for patients [166,206].
We briefly discuss efforts that use knowledge graphs to accomplish such tasks.
In contrast with most applications where node2vec and autoencoder models have become established, this field have focused on using graph attention models [207].
These models mimic machine translation models [208] and aim to simultaneously represent knowledge graphs in a low dimensional space and perform the task at hand.
Choi et al. used a graph attention model to predict patient diagnoses [126].
The authors constructed a directed graph using medical concepts from patient EHR data.
This directed graph was fed into a graph attention network and then used to predict a patient’s likelihood of heart failure [126].
Other approaches have used graph attention models to perform clinical tasks such as drug safety recommendations [167] and patient diagnoses [209].
Knowledge graphs have shown promising results when used for clinical applications; however, there is still room for improvement.
Most approaches have run into the common problem of missing data within EHR [126,166,167].
Future directions for the field consist of designing algorithms that can fill in this missing data gap or construct models that can take missing data into account.
Knowledge graphs are becoming widely used in biomedicine, and we expect their use to continue to grow.
At the moment, most are constructed from databases derived from manual curation or from co-occurrences in text.
We expect that machine learning approaches will play a key role in quickly deriving new findings from these graphs.
Representing these knowledge graphs in a low dimensional space that captures a graph’s local and global structure can enable many downstream machine learning analyses, and methods to capture this structure are an active area of research.
As with any field, rigorous evaluation that can identify key factors that drive success is critical to moving the field forward.
In regard to knowledge graphs, evaluation remains difficult.
Experiments in this context require a significant amount of time and consequently resources [127,168].
Moving from open ended and uncontrolled evaluations that consist of describing findings that are consistent with the literature to blinded evaluations of the literature that corroborate predictions and non-predictions would be a valuable first step.
There are also well-documented biases related to node degree and degree distribution that must be considered for accurate evaluation [210].
Furthermore, the diversity of applications hinders the development of a standardized set of expected evaluations.
We anticipate that a fruitful avenue of research will be techniques that can produce low dimensional representations of knowledge graphs which distinguish between multiple node and edge types.
There are many different sources of bias that lead to spurious edges or incompleteness, and modeling these biases may support better representations of knowledge graphs.
It is a promising time for research into the construction and application of knowledge graphs.
The peer reviewed literature is growing at an increasing rate and maintaining a complete understanding is becoming increasingly challenging for scientists.
One path that scientists can take to maintain awareness is to become hyper-focused on specific areas of knowledge graph literature.
If advances in how these graphs are constructed, represented and applied can enable the linking of fields, we may be able to savor the benefits of this detailed knowledge without losing the broader contextual links.
3. Open Question Answering with Weakly Supervised Embedding Models
Antoine Bordes, Jason Weston, Nicolas Usunier
arXiv (2014-04-17) https://arxiv.org/abs/1404.4326
4. Neural Network-based Question Answering over Knowledge Graphs on Word and Character Level
Denis Lukovnikov, Asja Fischer, Jens Lehmann, Sören Auer
Association for Computing Machinery (ACM) (2017) https://doi.org/gfv8hp
DOI: 10.1145/3038912.3052675
6. PhenoGeneRanker: A Tool for Gene Prioritization Using Complete Multiplex Heterogeneous Networks
Cagatay Dursun, Naoki Shimoyama, Mary Shimoyama, Michael Schläppi, Serdar Bozdag
bioRxiv (2019-05-27) https://doi.org/gf6rtr
DOI: 10.1101/651000
7. Biological Random Walks: Integrating heterogeneous data in disease gene prioritization
Michele Gentili, Leonardo Martini, Manuela Petti, Lorenzo Farina, Luca Becchetti
Institute of Electrical and Electronics Engineers (IEEE) (2019-07) https://doi.org/gf6rts
DOI: 10.1109/cibcb.2019.8791472
9. Systematic integration of biomedical knowledge prioritizes drugs for repurposing
Daniel Scott Himmelstein, Antoine Lizee, Christine Hessler, Leo Brueggeman, Sabrina L Chen, Dexter Hadley, Ari Green, Pouya Khankhanian, Sergio E Baranzini
eLife (2017-09-22) https://doi.org/cdfk
DOI: 10.7554/elife.26726 · PMID: 28936969 · PMCID: PMC5640425
11. Towards a definition of knowledge graphs
Lisa Ehrlinger, Wolfram Wöß
SEMANTiCS (2016)
15. Privacy Inference on Knowledge Graphs: Hardness and Approximation
Jianwei Qian, Shaojie Tang, Huiqi Liu, Taeho Jung, Xiang-Yang Li
Institute of Electrical and Electronics Engineers (IEEE) (2016-12) https://doi.org/ggtjgz
DOI: 10.1109/msn.2016.030
21. DrugBank 5.0: a major update to the DrugBank database for 2018
David S Wishart, Yannick D Feunang, An C Guo, Elvis J Lo, Ana Marcu, Jason R Grant, Tanvir Sajed, Daniel Johnson, Carin Li, Zinat Sayeeda, … Michael Wilson
Nucleic Acids Research (2018-01-04) https://doi.org/gcwtzk
DOI: 10.1093/nar/gkx1037 · PMID: 29126136 · PMCID: PMC5753335
22. A network integration approach for drug-target interaction prediction and computational drug repositioning from heterogeneous information
Yunan Luo, Xinbin Zhao, Jingtian Zhou, Jinglin Yang, Yanqing Zhang, Wenhua Kuang, Jian Peng, Ligong Chen, Jianyang Zeng
Nature Communications (2017-09-18) https://doi.org/gbxwrc
DOI: 10.1038/s41467-017-00680-8 · PMID: 28924171 · PMCID: PMC5603535
25. Bio2RDF: Towards a mashup to build bioinformatics knowledge systems
François Belleau, Marc-Alexandre Nolin, Nicole Tourigny, Philippe Rigault, Jean Morissette
Journal of Biomedical Informatics (2008-10) https://doi.org/frqkq5
DOI: 10.1016/j.jbi.2008.03.004 · PMID: 18472304
27. Constructing biomedical domain-specific knowledge graph with minimum supervision
Jianbo Yuan, Zhiwei Jin, Han Guo, Hongxia Jin, Xianchao Zhang, Tristram Smith, Jiebo Luo
Knowledge and Information Systems (2019-03-23) https://doi.org/gf6v26
DOI: 10.1007/s10115-019-01351-4
28. Feature assisted stacked attentive shortest dependency path based Bi-LSTM model for protein–protein interaction
Shweta Yadav, Asif Ekbal, Sriparna Saha, Ankit Kumar, Pushpak Bhattacharyya
Knowledge-Based Systems (2019-02) https://doi.org/gf4788
DOI: 10.1016/j.knosys.2018.11.020
29. Biological Databases- Integration of Life Science Data
Nishant Toomula, Arun Kumar, Sathish Kumar D, Vijaya Shanti Bheemidi
Journal of Computer Science & Systems Biology (2012) https://doi.org/gf8qcb
DOI: 10.4172/jcsb.1000081
30. COSMIC: somatic cancer genetics at high-resolution
Simon A. Forbes, David Beare, Harry Boutselakis, Sally Bamford, Nidhi Bindal, John Tate, Charlotte G. Cole, Sari Ward, Elisabeth Dawson, Laura Ponting, … Peter J. Campbell
Nucleic Acids Research (2017-01-04) https://doi.org/f9v865
DOI: 10.1093/nar/gkw1121 · PMID: 27899578 · PMCID: PMC5210583
31. COSMIC: the Catalogue Of Somatic Mutations In Cancer
John G Tate, Sally Bamford, Harry C Jubb, Zbyslaw Sondka, David M Beare, Nidhi Bindal, Harry Boutselakis, Charlotte G Cole, Celestino Creatore, Elisabeth Dawson, … Simon A Forbes
Nucleic Acids Research (2019-01-08) https://doi.org/gf9hxg
DOI: 10.1093/nar/gky1015 · PMID: 30371878 · PMCID: PMC6323903
32. Recurated protein interaction datasets
Lukasz Salwinski, Luana Licata, Andrew Winter, David Thorneycroft, Jyoti Khadake, Arnaud Ceol, Andrew Chatr Aryamontri, Rose Oughtred, Michael Livstone, Lorrie Boucher, … Henning Hermjakob
Nature Methods (2009-12) https://doi.org/fgvkmf
DOI: 10.1038/nmeth1209-860 · PMID: 19935838
33. Literature-curated protein interaction datasets
Michael E Cusick, Haiyuan Yu, Alex Smolyar, Kavitha Venkatesan, Anne-Ruxandra Carvunis, Nicolas Simonis, Jean-François Rual, Heather Borick, Pascal Braun, Matija Dreze, … Marc Vidal
Nature Methods (2008-12-30) https://doi.org/d4j62p
DOI: 10.1038/nmeth.1284 · PMID: 19116613 · PMCID: PMC2683745
34. Curation accuracy of model organism databases
I. M. Keseler, M. Skrzypek, D. Weerasinghe, A. Y. Chen, C. Fulcher, G.-W. Li, K. C. Lemmer, K. M. Mladinich, E. D. Chow, G. Sherlock, P. D. Karp
Database (2014-06-12) https://doi.org/gf63jz
DOI: 10.1093/database/bau058 · PMID: 24923819 · PMCID: PMC4207230
35. OMIM.org: Online Mendelian Inheritance in Man (OMIM®), an online catalog of human genes and genetic disorders
Joanna S. Amberger, Carol A. Bocchini, François Schiettecatte, Alan F. Scott, Ada Hamosh
Nucleic Acids Research (2015-01-28) https://doi.org/gf8qb6
DOI: 10.1093/nar/gku1205 · PMID: 25428349 · PMCID: PMC4383985
37. Text mining and expert curation to develop a database on psychiatric diseases and their genes
Alba Gutiérrez-Sacristán, Àlex Bravo, Marta Portero-Tresserra, Olga Valverde, Antonio Armario, M. C. Blanco-Gandía, Adriana Farré, Lierni Fernández-Ibarrondo, Francina Fonseca, Jesús Giraldo, … Laura I. Furlong
Database (2017) https://doi.org/gf8qb5
DOI: 10.1093/database/bax043 · PMID: 29220439 · PMCID: PMC5502359
44. Re-curation and rational enrichment of knowledge graphs in Biological Expression Language
Charles Tapley Hoyt, Daniel Domingo-Fernández, Rana Aldisi, Lingling Xu, Kristian Kolpeja, Sandra Spalek, Esther Wollert, John Bachman, Benjamin M Gyori, Patrick Greene, Martin Hofmann-Apitius
Database (2019) https://doi.org/gf7hm4
DOI: 10.1093/database/baz068 · PMID: 31225582 · PMCID: PMC6587072
45. LocText: relation extraction of protein localizations to assist database curation
Juan Miguel Cejuela, Shrikant Vinchurkar, Tatyana Goldberg, Madhukar Sollepura Prabhu Shankar, Ashish Baghudana, Aleksandar Bojchevski, Carsten Uhlig, André Ofner, Pandu Raharja-Liu, Lars Juhl Jensen, Burkhard Rost
BMC Bioinformatics (2018-01-17) https://doi.org/gf8qb9
DOI: 10.1186/s12859-018-2021-9 · PMID: 29343218 · PMCID: PMC5773052
46. Evaluating the impact of pre-annotation on annotation speed and potential bias: natural language processing gold standard development for clinical named entity recognition in clinical trial announcements
Todd Lingren, Louise Deleger, Katalin Molnar, Haijun Zhai, Jareen Meinzen-Derr, Megan Kaiser, Laura Stoutenborough, Qi Li, Imre Solti
Journal of the American Medical Informatics Association (2014-05) https://doi.org/f5zggh
DOI: 10.1136/amiajnl-2013-001837 · PMID: 24001514 · PMCID: PMC3994857
48. BioSimplify: an open source sentence simplification engine to improve recall in automatic biomedical information extraction.
Siddhartha Jonnalagadda, Graciela Gonzalez
AMIA … Annual Symposium proceedings. AMIA Symposium (2010-11-13) https://www.ncbi.nlm.nih.gov/pubmed/21346999
PMID: 21346999 · PMCID: PMC3041388
49. The EU-ADR corpus: Annotated drugs, diseases, targets, and their relationships
Erik M. van Mulligen, Annie Fourrier-Reglat, David Gurwitz, Mariam Molokhia, Ainhoa Nieto, Gianluca Trifiro, Jan A. Kors, Laura I. Furlong
Journal of Biomedical Informatics (2012-10) https://doi.org/f36vn6
DOI: 10.1016/j.jbi.2012.04.004 · PMID: 22554700
50. Comparative experiments on learning information extractors for proteins and their interactions
Razvan Bunescu, Ruifang Ge, Rohit J. Kate, Edward M. Marcotte, Raymond J. Mooney, Arun K. Ramani, Yuk Wah Wong
Artificial Intelligence in Medicine (2005-02) https://doi.org/dhztpn
DOI: 10.1016/j.artmed.2004.07.016 · PMID: 15811782
52. The BioGRID interaction database: 2013 update
Andrew Chatr-aryamontri, Bobby-Joe Breitkreutz, Sven Heinicke, Lorrie Boucher, Andrew Winter, Chris Stark, Julie Nixon, Lindsay Ramage, Nadine Kolas, Lara O’Donnell, … Mike Tyers
Nucleic Acids Research (2012-11-30) https://doi.org/f4jmz4
DOI: 10.1093/nar/gks1158 · PMID: 23203989 · PMCID: PMC3531226
53. The Comparative Toxicogenomics Database: update 2019
Allan Peter Davis, Cynthia J Grondin, Robin J Johnson, Daniela Sciaky, Roy McMorran, Jolene Wiegers, Thomas C Wiegers, Carolyn J Mattingly
Nucleic Acids Research (2019-01-08) https://doi.org/gf8qb7
DOI: 10.1093/nar/gky868 · PMID: 30247620 · PMCID: PMC6323936
54. CARD 2017: expansion and model-centric curation of the comprehensive antibiotic resistance database
Baofeng Jia, Amogelang R. Raphenya, Brian Alcock, Nicholas Waglechner, Peiyao Guo, Kara K. Tsang, Briony A. Lago, Biren M. Dave, Sheldon Pereira, Arjun N. Sharma, … Andrew G. McArthur
Nucleic Acids Research (2017-01-04) https://doi.org/f9wbjs
DOI: 10.1093/nar/gkw1004 · PMID: 27789705 · PMCID: PMC5210516
63. BELMiner: adapting a rule-based relation extraction system to extract biological expression language statements from bio-medical literature evidence sentences
K. E. Ravikumar, Majid Rastegar-Mojarad, Hongfang Liu
Database (2017) https://doi.org/gf7rbx
DOI: 10.1093/database/baw156 · PMID: 28365720 · PMCID: PMC5467463
67. LimTox: a web tool for applied text mining of adverse event and toxicity associations of compounds, drugs and genes
Andres Cañada, Salvador Capella-Gutierrez, Obdulia Rabal, Julen Oyarzabal, Alfonso Valencia, Martin Krallinger
Nucleic Acids Research (2017-07-03) https://doi.org/gf479h
DOI: 10.1093/nar/gkx462 · PMID: 28531339 · PMCID: PMC5570141
71. RLIMS-P 2.0: A Generalizable Rule-Based Information Extraction System for Literature Mining of Protein Phosphorylation Information
Manabu Torii, Cecilia N. Arighi, Gang Li, Qinghua Wang, Cathy H. Wu, K. Vijay-Shanker
IEEE/ACM Transactions on Computational Biology and Bioinformatics (2015-01-01) https://doi.org/gf8fpv
DOI: 10.1109/tcbb.2014.2372765 · PMID: 26357075 · PMCID: PMC4568560
73. PhpSyntaxTree tool
A Eisenbach, M Eisenbach
(2006)
74. Spacy 2: Natural language understanding with bloom embeddings, convolutional neural networks and incremental parsing
Matthew Honnibal, Ines Montani
To appear (2017)
75. STRING v9.1: protein-protein interaction networks, with increased coverage and integration
Andrea Franceschini, Damian Szklarczyk, Sune Frankild, Michael Kuhn, Milan Simonovic, Alexander Roth, Jianyi Lin, Pablo Minguez, Peer Bork, Christian von Mering, Lars J. Jensen
Nucleic Acids Research (2012-11-29) https://doi.org/gf5kcd
DOI: 10.1093/nar/gks1094 · PMID: 23203871 · PMCID: PMC3531103
76. A comprehensive and quantitative comparison of text-mining in 15 million full-text articles versus their corresponding abstracts
David Westergaard, Hans-Henrik Stærfeldt, Christian Tønsberg, Lars Juhl Jensen, Søren Brunak
PLOS Computational Biology (2018-02-15) https://doi.org/gcx747
DOI: 10.1371/journal.pcbi.1005962 · PMID: 29447159 · PMCID: PMC5831415
77. STITCH 4: integration of protein–chemical interactions with user data
Michael Kuhn, Damian Szklarczyk, Sune Pletscher-Frankild, Thomas H. Blicher, Christian von Mering, Lars J. Jensen, Peer Bork
Nucleic Acids Research (2014-01) https://doi.org/f5shb4
DOI: 10.1093/nar/gkt1207 · PMID: 24293645 · PMCID: PMC3964996
80. A new method for prioritizing drug repositioning candidates extracted by literature-based discovery
Majid Rastegar-Mojarad, Ravikumar Komandur Elayavilli, Dingcheng Li, Rashmi Prasad, Hongfang Liu
Institute of Electrical and Electronics Engineers (IEEE) (2015-11) https://doi.org/gf479j
DOI: 10.1109/bibm.2015.7359766
82. STRING v10: protein–protein interaction networks, integrated over the tree of life
Damian Szklarczyk, Andrea Franceschini, Stefan Wyder, Kristoffer Forslund, Davide Heller, Jaime Huerta-Cepas, Milan Simonovic, Alexander Roth, Alberto Santos, Kalliopi P. Tsafou, … Christian von Mering
Nucleic Acids Research (2015-01-28) https://doi.org/f64rfn
DOI: 10.1093/nar/gku1003 · PMID: 25352553 · PMCID: PMC4383874
85. BioCreative V CDR task corpus: a resource for chemical disease relation extraction
Jiao Li, Yueping Sun, Robin J. Johnson, Daniela Sciaky, Chih-Hsuan Wei, Robert Leaman, Allan Peter Davis, Carolyn J. Mattingly, Thomas C. Wiegers, Zhiyong Lu
Database (2016-05-09) https://doi.org/gf5hfw
DOI: 10.1093/database/baw068 · PMID: 27161011 · PMCID: PMC4860626
90. Extraction of relations between genes and diseases from text and large-scale data analysis: implications for translational research
Àlex Bravo, Janet Piñero, Núria Queralt-Rosinach, Michael Rautschka, Laura I Furlong
BMC Bioinformatics (2015-02-21) https://doi.org/f7kn8s
DOI: 10.1186/s12859-015-0472-9 · PMID: 25886734 · PMCID: PMC4466840
94. Deep learning for pharmacovigilance: recurrent neural network architectures for labeling adverse drug reactions in Twitter posts
Anne Cocos, Alexander G Fiks, Aaron J Masino
Journal of the American Medical Informatics Association (2017-07) https://doi.org/gbp9nj
DOI: 10.1093/jamia/ocw180 · PMID: 28339747
95. Cross-Sentence N-ary Relation Extraction with Graph LSTMs
Nanyun Peng, Hoifung Poon, Chris Quirk, Kristina Toutanova, Wen-tau Yih
arXiv (2017-08-15) https://arxiv.org/abs/1708.03743
101. On the difficulty of training Recurrent Neural Networks
Razvan Pascanu, Tomas Mikolov, Yoshua Bengio
arXiv (2013-02-19) https://arxiv.org/abs/1211.5063
102. Revisiting Unreasonable Effectiveness of Data in Deep Learning Era
Chen Sun, Abhinav Shrivastava, Saurabh Singh, Abhinav Gupta
arXiv (2017-08-07) https://arxiv.org/abs/1707.02968
103. Efficient Estimation of Word Representations in Vector Space
Tomas Mikolov, Kai Chen, Greg Corrado, Jeffrey Dean
arXiv (2013-09-10) https://arxiv.org/abs/1301.3781
104. Distributed Representations of Words and Phrases and their Compositionality
Tomas Mikolov, Ilya Sutskever, Kai Chen, Greg Corrado, Jeffrey Dean
arXiv (2013-10-18) https://arxiv.org/abs/1310.4546
107. Extraction of protein–protein interactions (PPIs) from the literature by deep convolutional neural networks with various feature embeddings
Sung-Pil Choi
Journal of Information Science (2016-11-01) https://doi.org/gcv8bn
DOI: 10.1177/0165551516673485
109. Expanding a Database-derived Biomedical Knowledge Graph via Multi-relation Extraction from Biomedical Abstracts
David N. Nicholson, Daniel S. Himmelstein, Casey S. Greene
bioRxiv (2020-01-31) https://doi.org/gf6qxh
DOI: 10.1101/730085
110. Distant supervision for relation extraction without labeled data
Mike Mintz, Steven Bills, Rion Snow, Dan Jurafsky
Association for Computational Linguistics (ACL) (2009) https://doi.org/fg9q43
DOI: 10.3115/1690219.1690287
118. Learning protein protein interaction extraction using distant supervision
Philippe Thomas, Illés Solt, Roman Klinger, Ulf Leser
(2011-01)
119. Robust Distant Supervision Relation Extraction via Deep Reinforcement Learning
Pengda Qin, Weiran Xu, William Yang Wang
arXiv (2018-05-28) https://arxiv.org/abs/1805.09927
120. DSGAN: Generative Adversarial Training for Distant Supervision Relation Extraction
Pengda Qin, Weiran Xu, William Yang Wang
arXiv (2018-05-28) https://arxiv.org/abs/1805.09929
121. Noise Reduction Methods for Distantly Supervised Biomedical Relation Extraction
Gang Li, Cathy Wu, K. Vijay-Shanker
Association for Computational Linguistics (ACL) (2017) https://doi.org/ggmk8s
DOI: 10.18653/v1/w17-2323
123. Learning language in logic - genic interaction extraction challenge
C. Nédellec
Proceedings of the learning language in logic 2005 workshop at the international conference on machine learning (2005)
125. Concept annotation in the CRAFT corpus
Michael Bada, Miriam Eckert, Donald Evans, Kristin Garcia, Krista Shipley, Dmitry Sitnikov, William A Baumgartner, K Bretonnel Cohen, Karin Verspoor, Judith A Blake, Lawrence E Hunter
BMC Bioinformatics (2012-07-09) https://doi.org/gb8vdr
DOI: 10.1186/1471-2105-13-161 · PMID: 22776079 · PMCID: PMC3476437
126. GRAM: Graph-based Attention Model for Healthcare Representation Learning
Edward Choi, Mohammad Taha Bahadori, Le Song, Walter F. Stewart, Jimeng Sun
arXiv (2017-04-04) https://arxiv.org/abs/1611.07012
127. miRNA-Disease Association Prediction with Collaborative Matrix Factorization
Zhen Shen, You-Hua Zhang, Kyungsook Han, Asoke K. Nandi, Barry Honig, De-Shuang Huang
Complexity (2017) https://doi.org/ggmrpm
DOI: 10.1155/2017/2498957
129. Representation Learning on Graphs: Methods and Applications
William L. Hamilton, Rex Ying, Jure Leskovec
arXiv (2018-04-11) https://arxiv.org/abs/1709.05584
133. Translating embeddings for modeling multi-relational data
Antoine Bordes, Nicolas Usunier, Alberto García-Durán, Jason Weston, Oksana Yakhnenko
NIPS (2013)
141. Graph embedding on biomedical networks: methods, applications and evaluations
Xiang Yue, Zhen Wang, Jingong Huang, Srinivasan Parthasarathy, Soheil Moosavinasab, Yungui Huang, Simon M Lin, Wen Zhang, Ping Zhang, Huan Sun
Bioinformatics (2019-10-04) https://doi.org/ggmzpf
DOI: 10.1093/bioinformatics/btz718 · PMID: 31584634
146. A Comparison of Semantic Similarity Methods for Maximum Human Interpretability
Pinky Sitikhu, Kritish Pahi, Pujan Thapa, Subarna Shakya
arXiv (2019-11-01) https://arxiv.org/abs/1910.09129
149. PrTransH: Embedding Probabilistic Medical Knowledge from Real World EMR Data
Linfeng Li, Peng Wang, Yao Wang, Jinpeng Jiang, Buzhou Tang, Jun Yan, Shenghui Wang, Yuting Liu
arXiv (2019-09-04) https://arxiv.org/abs/1909.00672
154. edge2vec: Representation learning using edge semantics for biomedical knowledge discovery
Zheng Gao, Gang Fu, Chunping Ouyang, Satoshi Tsutsui, Xiaozhong Liu, Jeremy Yang, Christopher Gessner, Brian Foote, David Wild, Qi Yu, Ying Ding
arXiv (2019-05-29) https://arxiv.org/abs/1809.02269
155. Learning Graph Embeddings from WordNet-based Similarity Measures
Andrey Kutuzov, Mohammad Dorgham, Oleksiy Oliynyk, Chris Biemann, Alexander Panchenko
arXiv (2019-04-15) https://arxiv.org/abs/1808.05611
158. Adversarially Regularized Graph Autoencoder for Graph Embedding
Shirui Pan, Ruiqi Hu, Guodong Long, Jing Jiang, Lina Yao, Chengqi Zhang
arXiv (2019-01-09) https://arxiv.org/abs/1802.04407
160. Autoencoders, unsupervised learning and deep architectures
Pierre Baldi
Proceedings of the 2011 international conference on unsupervised and transfer learning workshop - volume 27 (2011)
162. GraphVAE: Towards Generation of Small Graphs Using Variational Autoencoders
Martin Simonovsky, Nikos Komodakis
arXiv (2018-02-13) https://arxiv.org/abs/1802.03480
166. Safe Medicine Recommendation via Medical Knowledge Graph Embedding
Meng Wang, Mengyue Liu, Jun Liu, Sen Wang, Guodong Long, Buyue Qian
arXiv (2017-10-27) https://arxiv.org/abs/1710.05980
167. GAMENet: Graph Augmented MEmory Networks for Recommending Medication Combination
Junyuan Shang, Cao Xiao, Tengfei Ma, Hongyan Li, Jimeng Sun
Proceedings of the AAAI Conference on Artificial Intelligence (2019-07-17) https://doi.org/ggkm7r
DOI: 10.1609/aaai.v33i01.33011126
168. Heterogeneous network embedding for identifying symptom candidate genes
Kuo Yang, Ning Wang, Guangming Liu, Ruyu Wang, Jian Yu, Runshun Zhang, Jianxin Chen, Xuezhong Zhou
Journal of the American Medical Informatics Association (2018-11) https://doi.org/gfg6nr
DOI: 10.1093/jamia/ocy117 · PMID: 30357378
169. Predicting Protein–Protein Interactions from Multimodal Biological Data Sources via Nonnegative Matrix Tri-Factorization
Hua Wang, Heng Huang, Chris Ding, Feiping Nie
Journal of Computational Biology (2013-04) https://doi.org/f4thrx
DOI: 10.1089/cmb.2012.0273 · PMID: 23509857
171. HMDD v3.0: a database for experimentally supported human microRNA–disease associations
Zhou Huang, Jiangcheng Shi, Yuanxu Gao, Chunmei Cui, Shan Zhang, Jianwei Li, Yuan Zhou, Qinghua Cui
Nucleic Acids Research (2019-01-08) https://doi.org/ggmrph
DOI: 10.1093/nar/gky1010 · PMID: 30364956 · PMCID: PMC6323994
175. LWPCMF: Logistic Weighted Profile-based Collaborative Matrix Factorization for Predicting MiRNA-Disease Associations
Meng-Meng Yin, Zhen Cui, Ming-Ming Gao, Jin-Xing Liu, Ying-Lian Gao
IEEE/ACM Transactions on Computational Biology and Bioinformatics (2019) https://doi.org/ggmrpk
DOI: 10.1109/tcbb.2019.2937774 · PMID: 31478868
176. Protein-protein interaction prediction via Collective Matrix Factorization
Qian Xu, Evan Wei Xiang, Qiang Yang
Institute of Electrical and Electronics Engineers (IEEE) (2010-12) https://doi.org/csnv5m
DOI: 10.1109/bibm.2010.5706537
177. A network embedding model for pathogenic genes prediction by multi-path random walking on heterogeneous network
Bo Xu, Yu Liu, Shuo Yu, Lei Wang, Jie Dong, Hongfei Lin, Zhihao Yang, Jian Wang, Feng Xia
BMC Medical Genomics (2019-12-23) https://doi.org/ggmrpq
DOI: 10.1186/s12920-019-0627-z · PMID: 31865919 · PMCID: PMC6927107
178. Predicting gene-disease associations from the heterogeneous network using graph embedding
Xiaochan Wang, Yuchong Gong, Jing Yi, Wen Zhang
Institute of Electrical and Electronics Engineers (IEEE) (2019-11) https://doi.org/ggmrpj
DOI: 10.1109/bibm47256.2019.8983134
182. Detection of protein complexes from multiple protein interaction networks using graph embedding
Xiaoxia Liu, Zhihao Yang, Shengtian Sang, Hongfei Lin, Jian Wang, Bo Xu
Artificial Intelligence in Medicine (2019-05) https://doi.org/ggmrpf
DOI: 10.1016/j.artmed.2019.04.001 · PMID: 31164203
183. Large-scale structural and textual similarity-based mining of knowledge graph to predict drug–drug interactions
Ibrahim Abdelaziz, Achille Fokoue, Oktie Hassanzadeh, Ping Zhang, Mohammad Sadoghi
Journal of Web Semantics (2017-05) https://doi.org/gcrwk3
DOI: 10.1016/j.websem.2017.06.002
184. Matrix Factorization-Based Prediction of Novel Drug Indications by Integrating Genomic Space
Wen Dai, Xi Liu, Yibo Gao, Lin Chen, Jianglong Song, Di Chen, Kuo Gao, Yongshi Jiang, Yiping Yang, Jianxin Chen, Peng Lu
Computational and Mathematical Methods in Medicine (2015) https://doi.org/gb58g8
DOI: 10.1155/2015/275045 · PMID: 26078775 · PMCID: PMC4452507
186. Drug-Target Interaction Prediction with Graph Regularized Matrix Factorization
Ali Ezzat, Peilin Zhao, Min Wu, Xiao-Li Li, Chee-Keong Kwoh
IEEE/ACM Transactions on Computational Biology and Bioinformatics (2017-05-01) https://doi.org/ggmrrp
DOI: 10.1109/tcbb.2016.2530062 · PMID: 26890921
188. Network-based prediction of drug–target interactions using an arbitrary-order proximity embedded deep forest
Xiangxiang Zeng, Siyi Zhu, Yuan Hou, Pengyue Zhang, Lang Li, Jing Li, L Frank Huang, Stephen J Lewis, Ruth Nussinov, Feixiong Cheng
Bioinformatics (2020-01-23) https://doi.org/ggmrrk
DOI: 10.1093/bioinformatics/btaa010 · PMID: 31971579 · PMCID: PMC7203727
189. DrPOCS: Drug Repositioning Based on Projection Onto Convex Sets
Yin-Ying Wang, Chunfeng Cui, Liqun Qi, Hong Yan, Xing-Ming Zhao
IEEE/ACM Transactions on Computational Biology and Bioinformatics (2019-01-01) https://doi.org/ggmrrq
DOI: 10.1109/tcbb.2018.2830384 · PMID: 29993698
192. Drug–Disease Association and Drug-Repositioning Predictions in Complex Diseases Using Causal Inference–Probabilistic Matrix Factorization
Jihong Yang, Zheng Li, Xiaohui Fan, Yiyu Cheng
Journal of Chemical Information and Modeling (2014-08-22) https://doi.org/f6hpb4
DOI: 10.1021/ci500340n · PMID: 25116798
195. Scalable and Accurate Drug–target Prediction Based on Heterogeneous Bio-linked Network Mining
Nansu Zong, Rachael Sze Nga Wong, Victoria Ngo, Yue Yu, Ning Li
bioRxiv (2019-02-03) https://doi.org/ggmrrm
DOI: 10.1101/539643
196. Drug Similarity Integration Through Attentive Multi-view Graph
Auto-Encoders
Tengfei Ma, Cao Xiao, Jiayu Zhou, Fei Wang
arXiv (2018-04-28) https://arxiv.org/abs/1804.10850v1
198. DrugBank: a knowledgebase for drugs, drug actions and drug targets
David S. Wishart, Craig Knox, An Chi Guo, Dean Cheng, Savita Shrivastava, Dan Tzur, Bijaya Gautam, Murtaza Hassanali
Nucleic Acids Research (2008-01-01) https://doi.org/d3qqpj
DOI: 10.1093/nar/gkm958 · PMID: 18048412 · PMCID: PMC2238889
201. Drug-Drug Interaction Prediction Based on Knowledge Graph Embeddings and Convolutional-LSTM Network
Md. Rezaul Karim, Michael Cochez, Joao Bosco Jares, Mamtaz Uddin, Oya Beyan, Stefan Decker
Association for Computing Machinery (ACM) (2019-09-04) https://doi.org/ggmrrs
DOI: 10.1145/3307339.3342161
203. Applying linked data principles to represent patient’s electronic health records at Mayo clinic
Jyotishman Pathak, Richard C. Kiefer, Christopher G. Chute
Association for Computing Machinery (ACM) (2012) https://doi.org/fzm2p7
DOI: 10.1145/2110363.2110415
204. PDD Graph: Bridging Electronic Medical Records and Biomedical Knowledge Graphs via Entity Linking
Meng Wang, Jiaheng Zhang, Jun Liu, Wei Hu, Sen Wang, Xue Li, Wenqiang Liu
arXiv (2017-07-25) https://arxiv.org/abs/1707.05340
205. Diagnosis Code Assignment Using Sparsity-Based Disease Correlation Embedding
Sen Wang, Xiaojun Chang, Xue Li, Guodong Long, Lina Yao, Quan Z. Sheng
IEEE Transactions on Knowledge and Data Engineering (2016-12-01) https://doi.org/f9cgtv
DOI: 10.1109/tkde.2016.2605687
206. EMR-based medical knowledge representation and inference via Markov random fields and distributed representation learning
Chao Zhao, Jingchi Jiang, Yi Guan
arXiv (2017-09-21) https://arxiv.org/abs/1709.06908
207. Attention Is All You Need
Ashish Vaswani, Noam Shazeer, Niki Parmar, Jakob Uszkoreit, Llion Jones, Aidan N. Gomez, Lukasz Kaiser, Illia Polosukhin
arXiv (2017-12-07) https://arxiv.org/abs/1706.03762
208. Neural Machine Translation by Jointly Learning to Align and Translate
Dzmitry Bahdanau, Kyunghyun Cho, Yoshua Bengio
arXiv (2016-05-23) https://arxiv.org/abs/1409.0473
209. Learning the Graphical Structure of Electronic Health Records with Graph Convolutional Transformer
Edward Choi, Zhen Xu, Yujia Li, Michael W. Dusenberry, Gerardo Flores, Yuan Xue, Andrew M. Dai
arXiv (2020-01-22) https://arxiv.org/abs/1906.04716
210. The probability of edge existence due to node degree: a baseline for network-based predictions
Michael Zietz, Daniel S. Himmelstein, Kyle Kloster, Christopher Williams, Michael W. Nagle, Blair D. Sullivan, Casey S. Greene
Manubot (2020-03-05) https://greenelab.github.io/xswap-manuscript/
0000-0003-0002-5761
·
danich1
0000-0001-8713-9213
·
cgreene
·
greenescientist